Chapter 1 Temperature and Heat
1.6 Mechanisms of Heat Transfer
OpenStax and Paula Herrera-Siklody
Learning Objectives
By the end of this section, you will be able to:
- Explain some phenomena that involve conductive, convective, and radiative heat transfer
- Solve problems on the relationships between heat transfer, time, and rate of heat transfer
- Solve problems using the formulas for conduction and radiation
Just as interesting as the effects of heat transfer on a system are the methods by which it occurs. Whenever there is a temperature difference, heat transfer occurs. It may occur rapidly, as through a cooking pan, or slowly, as through the walls of a picnic ice chest. So many processes involve heat transfer that it is hard to imagine a situation where no heat transfer occurs. Yet every heat transfer takes place by only three methods:
- Conduction is heat transfer through stationary matter by physical contact. (The matter is stationary on a macroscopic scale—we know that thermal motion of the atoms and molecules occurs at any temperature above absolute zero.) Heat transferred from the burner of a stove through the bottom of a pan to food in the pan is transferred by conduction.
- Convection is the heat transfer by the macroscopic movement of a fluid. This type of transfer takes place in a forced-air furnace and in weather systems, for example.
- Heat transfer by radiation occurs when microwaves, infrared radiation, visible light, or another form of electromagnetic radiation is emitted or absorbed. An obvious example is the warming of Earth by the Sun. A less obvious example is thermal radiation from the human body.
In the illustration at the beginning of this chapter, the fire warms the snowshoers’ faces largely by radiation. Convection carries some heat to them, but most of the air flow from the fire is upward (creating the familiar shape of flames), carrying heat to the food being cooked and into the sky. The snowshoers wear clothes des igned with low conductivity to prevent heat flow out of their bodies.
In this section, we examine these methods in some detail. Each method has unique and interesting characteristics, but all three have two things in common: They transfer heat solely because of a temperature difference, and the greater the temperature difference, the faster the heat transfer (Figure 1.19).
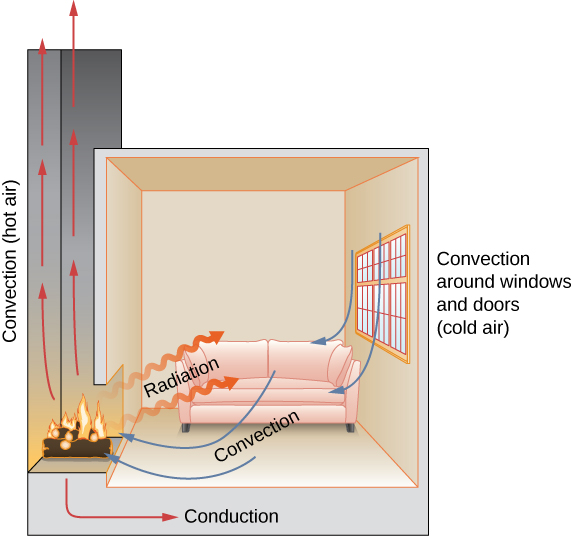
Check Your Understanding 1.6
Name an example from daily life (different from the text) for each mechanism of heat transfer.
Conduction
As you walk barefoot across the living room carpet in a cold house and then step onto the kitchen tile floor, your feet feel colder on the tile. This result is intriguing, since the carpet and tile floor are both at the same temperature. The different sensation is explained by the different rates of heat transfer: The heat loss is faster for skin in contact with the tiles than with the carpet, so the sensation of cold is more intense.
Some materials conduct thermal energy faster than others. Figure 1.20 shows a material that conducts heat slowly—it is a good thermal insulator, or poor heat conductor—used to reduce heat flow into and out of a house.

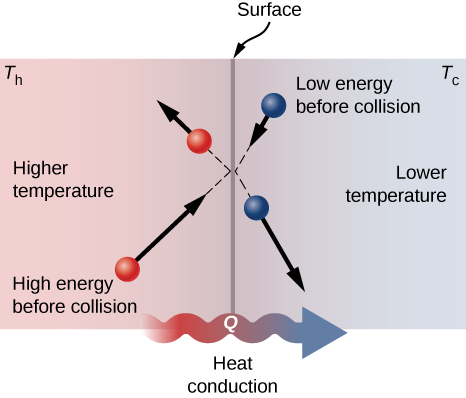
A molecular picture of heat conduction will help justify the equation that describes it. Figure 1.21 shows molecules in two bodies at different temperatures, [latex]T_\text{h}[/latex] and [latex]T_\text{c}[/latex], for “hot” and “cold.” The average kinetic energy of a molecule in the hot body is higher than in the colder body. If two molecules collide, energy transfers from the high-energy to the low-energy molecule. In a metal, the picture would also include free valence electrons colliding with each other and with atoms, likewise transferring energy. The cumulative effect of all collisions is a net flux of heat from the hotter body to the colder body. Thus, the rate of heat transfer increases with increasing temperature difference [latex]{\Delta}T = T_\text{h} -T_\text{c}[/latex]. If the temperatures are the same, the net heat transfer rate is zero. Because the number of collisions increases with the increasing area, heat conduction is proportional to the cross-sectional area—a second factor in the equation.
A third quantity that affects the conduction rate is the thickness of the material through which heat transfers. Figure 1.22 shows a slab of material with a higher temperature on the left than on the right. Heat transfers from the left to the right by a series of molecular collisions. The greater the distance between hot and cold, the more time the material takes to transfer the same amount of heat.
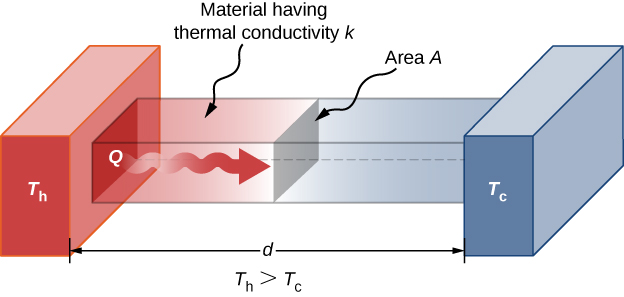
All four of these quantities appear in a simple equation deduced from and confirmed by experiments. The rate of conductive heat transfer through a slab of material, such as the one in Figure 1.22, is given by
[latex]\begin{equation} \tag{1.9} P = \frac{dQ}{dt} = \frac{kA(T_h - T_c)}{d} \end{equation}[/latex]
where P is the power or rate of heat transfer in watts or in kilocalories per second, A and d are its surface area and thickness, as shown in Figure 1.22, [latex]T_h - T_c[/latex] is the temperature difference across the slab, and k is the thermal conductivity of the material. Table 1.5 gives representative values of thermal conductivity.
More generally, we can write
[latex]P = -kA\frac{dT}{dx},[/latex]
where x is the coordinate in the direction of heat flow. Since in Figure 1.22, the power and area are constant, dT/dx is constant, and the temperature decreases linearly from [latex]T_h[/latex] to [latex]T_c[/latex].
| Substance | Thermal Conductivity k ([latex]W/m \cdot ^{\circ} \text C[/latex]) |
| Diamond | 2000 |
| Silver | 420 |
| Copper | 390 |
| Gold | 318 |
| Aluminum | 220 |
| Steel iron | 80 |
| Steel (stainless) | 14 |
| Ice | 2.2 |
| Glass (average) | 0.84 |
| Concrete Brick | 0.84 |
| Water | 0.6 |
| Fatty tissue (without blood) | 0.2 |
| Asbestos | 0.16 |
| Plasterboard | 0.16 |
| Wood | 0.08–0.16 |
| Snow (dry) | 0.10 |
| Cork | 0.042 |
| Glass wool | 0.042 |
| Wool | 0.04 |
| Down feathers | 0.025 |
| Air | 0.023 |
| Polystyrene foam | 0.010 |
Example 1.10
Calculating Heat Transfer through Conduction
A polystyrene foam icebox has a total area of [latex]0.950 m^2[/latex] and walls with an average thickness of 2.50 cm. The box contains ice, water, and canned beverages at [latex]0 ^{\circ}\text{C}[/latex]. The inside of the box is kept cold by melting ice. How much ice melts in one day if the icebox is kept in the trunk of a car at [latex]35.0 ^{\circ}\text{C}[/latex]?
Strategy
This question involves both heat for a phase change (melting of ice) and the transfer of heat by conduction. To find the amount of ice melted, we must find the net heat transferred. This value can be obtained by calculating the rate of heat transfer by conduction and multiplying by time.
Solution
First we identify the knowns.
[latex]k = 0.010 W/m\cdot^\circ C[/latex] for polystyrene foam; [latex]A = 0.950 m^2[/latex]; [latex]d = 2.50\text{cm} = 0.0250 \text{m}[/latex]; [latex]T_\text{c} = 0^\circ \text{C}[/latex]; [latex]T_\text{h} = 35.0^\circ \text{C}[/latex]; [latex]t = 1 \text{day} = 24 \text{hours}-86,400\text{s}.[/latex]
Then we identify the unknowns. We need to solve for the mass of the ice, m. We also need to solve for the net heat transferred to melt the ice, Q. The rate of heat transfer by conduction is given by
[latex]P = \frac{dQ}{dt} = \frac{kA(T_h - T_c)}{d}.[/latex]
The heat used to melt the ice is [latex]Q = mL_f[/latex]. We insert the known values:
[latex]P = \frac{(0.010\text{W/m}\cdot ^\circ \text{C})(0.950\text{m}^2)(35.0^\circ \text{C} - 0^\circ \text{C})}{0.0250 \text{m}} = 13.3 \text{W}.[/latex]
Multiplying the rate of heat transfer by the time we obtain
[latex]Q = Pt = (13.3 \text{W})(86.400\text{s}) = 1.15 \times 10^6 \text{J}.[/latex]
We set this equal to the heat transferred to melt the ice, [latex]Q=mL_f[/latex], and solve for the mass m:
[latex]m = \frac{Q}{L_\text{f}} = \frac{1.15 \times 10^6 \text{J}}{334 \times 10^3 \text{J/kg}} = 3.44kg.[/latex]
Significance
The result of 3.44 kg, or about 7.6 lb, seems about right, based on experience. You might expect to use about a 4 kg (7–10 lb) bag of ice per day. A little extra ice is required if you add any warm food or beverages.
Table 1.5 shows that polystyrene foam is a very poor conductor and thus a good insulator. Other good insulators include fiberglass, wool, and goose down feathers. Like polystyrene foam, these all contain many small pockets of air, taking advantage of air’s poor thermal conductivity.
In developing insulation, the smaller the conductivity k and the larger the thickness d, the better. Thus, the ratio d/k, called the R factor, is large for a good insulator. The rate of conductive heat transfer is inversely proportional to R. R factors are most commonly quoted for household insulation, refrigerators, and the like. Unfortunately, in the United States, R is still in non-metric units of [latex]\text{ft}^2 \cdot ^\circ \text{F} \cdot \text{h/Btu}[/latex], although the unit usually goes unstated [1 British thermal unit (Btu) is the amount of energy needed to change the temperature of 1.0 lb of water by [latex]1.0 ^{\circ}\text{F}[/latex], which is 1055.1 J]. A couple of representative values are an R factor of 11 for 3.5-inch-thick fiberglass batts (pieces) of insulation and an R factor of 19 for 6.5-inch-thick fiberglass batts (Figure 1.23). In the US, walls are usually insulated with 3.5-inch batts, whereas ceilings are usually insulated with 6.5-inch batts. In cold climates, thicker batts may be used.

Note that in Table 1.5, most of the best thermal conductors—silver, copper, gold, and aluminum—are also the best electrical conductors, because they contain many free electrons that can transport thermal energy. (Diamond, an electrical insulator, conducts heat by atomic vibrations.) Cooking utensils are typically made from good conductors, but the handles of those used on the stove are made from good insulators (bad conductors).
Example 1.11
Two Conductors End to End
A steel rod and an aluminum rod, each of diameter 1.00 cm and length 25.0 cm, are welded end to end. One end of the steel rod is placed in a large tank of boiling water at [latex]100^{\circ}\text{C}[/latex], while the far end of the aluminum rod is placed in a large tank of water at [latex]20^{\circ}\text{C}[/latex]. The rods are insulated so that no heat escapes from their surfaces. What is the temperature at the joint, and what is the rate of heat conduction through this composite rod?
Strategy
The heat that enters the steel rod from the boiling water has no place to go but through the steel rod, then through the aluminum rod, to the cold water. Therefore, we can equate the rate of conduction through the steel to the rate of conduction through the aluminum.
We repeat the calculation with a second method, in which we use the thermal resistance R of the rod, since it simply adds when two rods are joined end to end. (We will use a similar method in the chapter on direct-current circuits.)
Solution
- Identify the knowns and convert them to SI units.
The length of each rod is [latex]L_\text{A1} = L_\text{steel} = 0.25 \text{m}[/latex], the cross-sectional area of each rod is [latex]A_\text{A1} = A_\text{steel} = 7.85 \times 10^{-5}\text{m}^2[/latex], the thermal conductivity of aluminum is [latex]k_{A1}=220 \text{W}/m \cdot ^{\circ} \text{C}[/latex], the thermal conductivity of steel is [latex]k_\text{steel}=80 \text{W}/m \cdot ^{\circ} \text{C}[/latex], the temperature at the hot end is [latex]T = 100^{\circ}\text{C}[/latex], and the temperature at the cold end is [latex]T = 20^{\circ}\text{C}[/latex]. - Calculate the heat-conduction rate through the steel rod and the heat-conduction rate through the aluminum rod in terms of the unknown temperature T at the joint:
[latex]\begin{array}{rl}P_{steel} & = \frac{k_{steel} A_{steel} \Delta T_{steel}}{L_{steel}} \\ & = \frac{(80W/m \cdot ^{\circ} \text{C})(7.85 \times {10}^{-5} m^{2} )(100^{\circ}\text{C} - T)}{0.25m} \\ & = (0.0251W/^{\circ} \text{C})(100^{\circ} \text{C} - T);\end{array}[/latex]
[latex]\begin{array}{rl}P_{A1} & = \frac{k_{Al} A_{A1} \Delta T_{Al}}{L_{A1}} \\ &= \frac{(220W/m \cdot ^{\circ}\text{C})(7.85 \times {10}^{-5} m^{2} )(T - 20^{\circ}\text{C})}{0.25m} \\ &= (0.0691W/^{\circ}\text{C})(T - 20^{\circ}\text{C}) .\end{array}[/latex] - Set the two rates equal and solve for the unknown temperature:
[latex]\begin{array}{rl}(0.0691 \text{W}/^{\circ}\text{C})(T-20^{\circ}\text{C}) &= (0.0251\text{W}/^{\circ}\text{C})(100^{\circ}\text{C}-T)\\ T &= 41.3^{\circ}\text{C}.\end{array}[/latex] - Calculate either rate:
[latex]P_\text{steel} = (0.0251\text{W/}^\circ \text{C})(100^\circ \text{C} - 41.3^\circ \text{C}) = 1.47\text{W}.[/latex] - If desired, check your answer by calculating the other rate.
Solution
- Recall that [latex]R=L/k[/latex]. Now [latex]P=A\Delta T/R[/latex],or [latex]\Delta T = PR/A[/latex].
- We know that [latex]\Delta T_\text{steel} + \Delta T_\text{Al} = 100^\circ \text{C} - 20^\circ \text{C} = 80 ^\circ \text{C}[/latex]. We also know that [latex]P_\text{steel} = P_\text{Al}[/latex], and we denote that rate of heat flow by P. Combine the equations:
[latex]\frac{PR_\text{steel}}{A} + \frac{PR_\text{Al}}{A} = 80^\circ \text{C}.[/latex]
Thus, we can simply add R factors. Now, [latex]P= \frac{80^\circ \text{C}}{A(R_\text{steel}+R_\text{Al})}[/latex]. - Find the [latex]R_s[/latex] from the known quantities:
[latex]R_\text{steel} = 3.13 \times 10^{-3}\text{m}^2 \cdot ^\circ\text{C/W}[/latex]
and
[latex]R_\text{Al} = 1.14 \times 10^{-3}\text{m}^2\cdot ^\circ\text{C/W}[/latex] - Substitute these values in to find [latex]P=1.47\text{W}[/latex] as before.
- Determine [latex]\Delta T[/latex] for the aluminum rod (or for the steel rod) and use it to find T at the joint.
[latex]\Delta T_\text{Al} = \frac{PR_\text{Al}}{A} = \frac{(1.47\text{W})(1.14 \times 10^{-3}\text{m}^2\cdot ^\circ \text{C/W})}{7.85 \times 10^{-5}\text{m}^2} = 21.3^\circ \text{C},[/latex]
so [latex]T = 20^\circ \text{C} + 21.3^\circ \text{C} = 41.3^\circ \text{C}[/latex], as in Solution 1. - If desired, check by determining [latex]\Delta T[/latex] for the other rod.
Significance
In practice, adding R values is common, as in calculating the R value of an insulated wall. In the analogous situation in electronics, the resistance corresponds to AR in this problem and is additive even when the areas are unequal, as is common in electronics. Our equation for heat conduction can be used only when the areas are equal; otherwise, we would have a problem in three-dimensional heat flow, which is beyond our scope.
Check Your Understanding 1.7
How does the rate of heat transfer by conduction change when all spatial dimensions are doubled?
Conduction is caused by the random motion of atoms and molecules. As such, it is an ineffective mechanism for heat transport over macroscopic distances and short times. For example, the temperature on Earth would be unbearably cold during the night and extremely hot during the day if heat transport in the atmosphere were only through conduction. Also, car engines would overheat unless there was a more efficient way to remove excess heat from the pistons. The next module discusses the important heat-transfer mechanism in such situations.
Convection
In convection, thermal energy is carried by the large-scale flow of matter. It can be divided into two types. In forced convection, the flow is driven by fans, pumps, and the like. A simple example is a fan that blows air past you in hot surroundings and cools you by replacing the air heated by your body with cooler air. A more complicated example is the cooling system of a typical car, in which a pump moves coolant through the radiator and engine to cool the engine and a fan blows air to cool the radiator.
In free or natural convection, the flow is driven by buoyant forces: hot fluid rises and cold fluid sinks because density decreases as temperature increases. The house in Figure 1.24 is kept warm by natural convection, as is the pot of water on the stove in Figure 1.25. Ocean currents and large-scale atmospheric circulation, which result from the buoyancy of warm air and water, transfer hot air from the tropics toward the poles and cold air from the poles toward the tropics. (Earth’s rotation interacts with those flows, causing the observed eastward flow of air in the temperate zones.)
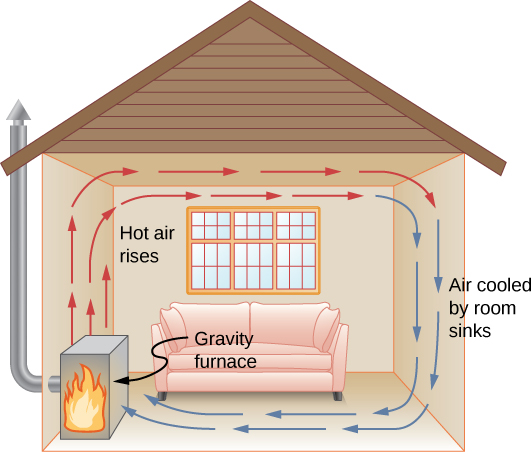
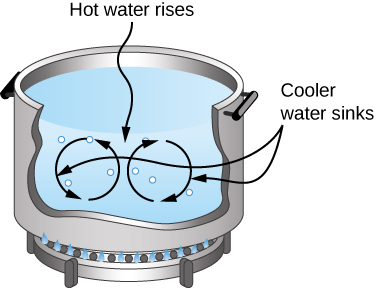
Interactive
Natural convection like that of Figure 1.24 and Figure 1.25, but acting on rock in Earth’s mantle, drives plate tectonics that are the motions that have shaped Earth’s surface.
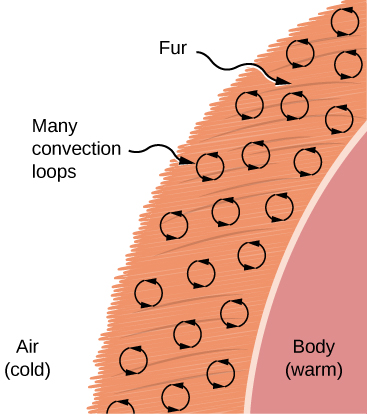
Convection is usually more complicated than conduction. Beyond noting that the convection rate is often approximately proportional to the temperature difference, we will not do any quantitative work comparable to the formula for conduction. However, we can describe convection qualitatively and relate convection rates to heat and time. Air is a poor conductor, so convection dominates heat transfer by air. Therefore, the amount of available space for airflow determines whether air transfers heat rapidly or slowly. There is little heat transfer in a space filled with air with a small amount of other material that prevents flow. The space between the inside and outside walls of a typical American house, for example, is about 9 cm (3.5 in.)—large enough for convection to work effectively. The addition of wall insulation prevents airflow, so heat loss (or gain) is decreased. On the other hand, the gap between the two panes of a double-paned window is about 1 cm, which largely prevents convection and takes advantage of air’s low conductivity reduce heat loss. Fur, cloth, and fiberglass also take advantage of the low conductivity of air by trapping it in spaces too small to support convection (Figure 1.26).
Some interesting phenomena happen when convection is accompanied by a phase change. The combination allows us to cool off by sweating even if the temperature of the surrounding air exceeds body temperature. Heat from the skin is required for sweat to evaporate from the skin, but without air flow, the air becomes saturated and evaporation stops. Air flow caused by convection replaces the saturated air by dry air and evaporation continues.
Example 1.12
Calculating the Flow of Mass during Convection
The average person produces heat at the rate of about 120 W when at rest. At what rate must water evaporate from the body to get rid of all this energy? (For simplicity, we assume this evaporation occurs when a person is sitting in the shade and surrounding temperatures are the same as skin temperature, eliminating heat transfer by other methods.)
Strategy
Energy is needed for this phase change [latex](Q =mL_v)[/latex]. Thus, the energy loss per unit time is
[latex]\frac{Q}{t} = \frac{mL_V}{t} = 120 \text{W} = 120 \text{J/s}.[/latex]
We divide both sides of the equation by [latex]L_v[/latex] to find that the mass evaporated per unit time is
[latex]\frac{m}{t} = \frac{120\text{J/s}}{L_V}.[/latex]
Solution
Insert the value of the latent heat from Table 1.4, [latex]L_v = 2430\text{kJ/kg} = 2430\text{J/g}[/latex]. This yields
[latex]\frac{m}{t} = \frac{120 \text{J/s}}{2430 \text{J/g}} = 0.0494 \text{g/s} =2.96 \text {g/min}.[/latex]
Significance
Evaporating about 3 g/min seems reasonable. This would be about 180 g (about 7 oz.) per hour. If the air is very dry, the sweat may evaporate without even being noticed. A significant amount of evaporation also takes place in the lungs and breathing passages.
Another important example of the combination of phase change and convection occurs when water evaporates from the oceans. Heat is removed from the ocean when water evaporates. If the water vapor condenses in liquid droplets as clouds form, possibly far from the ocean, heat is released in the atmosphere. Thus, there is an overall transfer of heat from the ocean to the atmosphere. This process is the driving power behind thunderheads, those great cumulus clouds that rise as much as 20.0 km into the stratosphere (Figure 1.27). Water vapor carried in by convection condenses, releasing tremendous amounts of energy. This energy causes the air to expand and rise to colder altitudes. More condensation occurs in these regions, which in turn drives the cloud even higher. This mechanism is an example of positive feedback, since the process reinforces and accelerates itself. It sometimes produces violent storms, with lightning and hail. The same mechanism drives hurricanes.
Interactive
This time-lapse video shows convection currents in a thunderstorm, including “rolling” motion similar to that of boiling water.

Check Your Understanding 1.8
Explain why using a fan in the summer feels refreshing.
Radiation
You can feel the heat transfer from the Sun. The space between Earth and the Sun is largely empty, so the Sun warms us without any possibility of heat transfer by convection or conduction. Similarly, you can sometimes tell that the oven is hot without touching its door or looking inside—it may just warm you as you walk by. In these examples, heat is transferred by radiation (Figure 1.28). That is, the hot body emits electromagnetic waves that are absorbed by the skin. No medium is required for electromagnetic waves to propagate. Different names are used for electromagnetic waves of different wavelengths: radio waves, microwaves, infrared radiation, visible light, ultraviolet radiation, X-rays, and gamma rays.

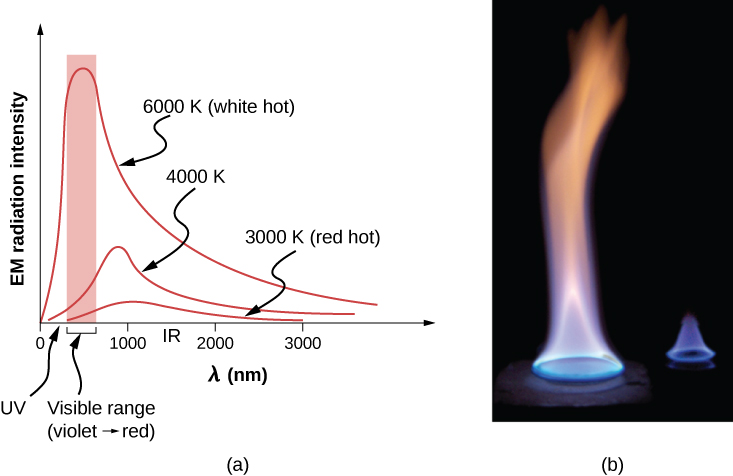
The energy of electromagnetic radiation varies over a wide range, depending on the wavelength: A shorter wavelength (or higher frequency) corresponds to a higher energy. Because more heat is radiated at higher temperatures, higher temperatures produce more intensity at every wavelength but especially at shorter wavelengths. In visible light, wavelength determines color—red has the longest wavelength and violet the shortest—so a temperature change is accompanied by a color change. For example, an electric heating element on a stove glows from red to orange, while the higher-temperature steel in a blast furnace glows from yellow to white. Infrared radiation is the predominant form radiated by objects cooler than the electric element and the steel. The radiated energy as a function of wavelength depends on its intensity, which is represented in Figure 1.29 by the height of the distribution. (Electromagnetic Waves explains more about the electromagnetic spectrum, and Photons and Matter Waves discusses why the decrease in wavelength corresponds to an increase in energy.)
The rate of heat transfer by radiation also depends on the object’s color. Black is the most effective, and white is the least effective. On a clear summer day, black asphalt in a parking lot is hotter than adjacent gray sidewalk, because black absorbs better than gray (Figure 1.30). The reverse is also true—black radiates better than gray. Thus, on a clear summer night, the asphalt is colder than the gray sidewalk, because black radiates the energy more rapidly than gray. A perfectly black object would be an ideal radiator and an ideal absorber, as it would capture all the radiation that falls on it. In contrast, a perfectly white object or a perfect mirror would reflect all radiation, and a perfectly transparent object would transmit it all (Figure 1.31). Such objects would not emit any radiation. Mathematically, the color is represented by the emissivity e. A “blackbody” radiator would have an [latex]e=1[/latex], whereas a perfect reflector or transmitter would have [latex]e=0[/latex]. For real examples, tungsten light bulb filaments have an e of about 0.5, and carbon black (a material used in printer toner) has an emissivity of about 0.95.

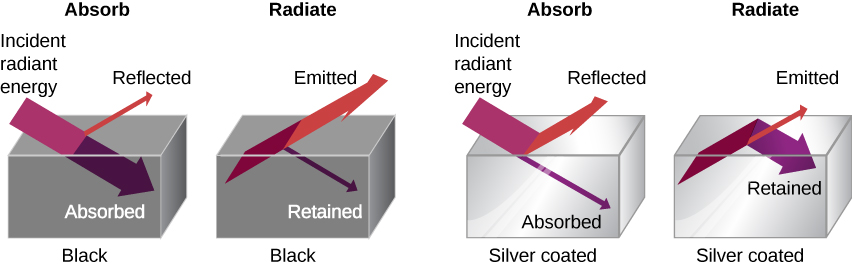
To see that, consider a silver object and a black object that can exchange heat by radiation and are in thermal equilibrium. We know from experience that they will stay in equilibrium (the result of a principle that will be discussed at length in Second Law of Thermodynamics). For the black object’s temperature to stay constant, it must emit as much radiation as it absorbs, so it must be as good at radiating as absorbing. Similar considerations show that the silver object must radiate as little as it absorbs. Thus, one property, emissivity, controls both radiation and absorption.
Finally, the radiated heat is proportional to the object’s surface area, since every part of the surface radiates. If you knock apart the coals of a fire, the radiation increases noticeably due to an increase in radiating surface area.
The rate of heat transfer by emitted radiation is described by the Stefan-Boltzmann law of radiation:
[latex]P = \sigma AeT^4,[/latex]
where [latex]\sigma = 5.67 \times 10^{-8}\text{J}/\text{s}\cdot m^2\cdot K^4[/latex] is the Stefan-Boltzmann constant, a combination of fundamental constants of nature; A is the surface area of the object; and T is its temperature in kelvins.
The proportionality to the fourth power of the absolute temperature is a remarkably strong temperature dependence. It allows the detection of even small temperature variations. Images called thermographs can be used medically to detect regions of abnormally high temperature in the body, perhaps indicative of disease. Similar techniques can be used to detect heat leaks in homes (Figure 1.32), optimize performance of blast furnaces, improve comfort levels in work environments, and even remotely map Earth’s temperature profile.

The Stefan-Boltzmann equation needs only slight refinement to deal with a simple case of an object’s absorption of radiation from its surroundings. Assuming that an object with a temperature [latex]T_1[/latex] is surrounded by an environment with uniform temperature [latex]T_2[/latex], the net rate of heat transfer by radiation is
\begin{equation} \tag{1.10}
P_\text{net} = \sigma eA(T_{2}^{4} – T_{1}^{4}),
\end{equation}
where e is the emissivity of the object alone. In other words, it does not matter whether the surroundings are white, gray, or black: The balance of radiation into and out of the object depends on how well it emits and absorbs radiation. When [latex]T_2 >T_1[/latex], the quantity [latex]P_\text{net}[/latex] is positive, that is, the net heat transfer is from hot to cold.
Before doing an example, we have a complication to discuss: different emissivities at different wavelengths. If the fraction of incident radiation an object reflects is the same at all visible wavelengths, the object is gray; if the fraction depends on the wavelength, the object has some other color. For instance, a red or reddish object reflects red light more strongly than other visible wavelengths. Because it absorbs less red, it radiates less red when hot. Differential reflection and absorption of wavelengths outside the visible range have no effect on what we see, but they may have physically important effects. Skin is a very good absorber and emitter of infrared radiation, having an emissivity of 0.97 in the infrared spectrum. Thus, in spite of the obvious variations in skin color, we are all nearly black in the infrared. This high infrared emissivity is why we can so easily feel radiation on our skin. It is also the basis for the effectiveness of night-vision scopes used by law enforcement and the military to detect human beings.
Example 1.13
Calculating the Net Heat Transfer of a Person
What is the rate of heat transfer by radiation of an unclothed person standing in a dark room whose ambient temperature is [latex]22.0^\circ \text{C}[/latex]? The person has a normal skin temperature of [latex]33.0^\circ \text{C}[/latex] and a surface area of [latex]1.50\text{m}^{2}[/latex]. The emissivity of skin is 0.97 in the infrared, the part of the spectrum where the radiation takes place.
Strategy
We can solve this by using the equation for the rate of radiative heat transfer.
Solution
Insert the temperature values [latex]T_2 = 295 \text{K}[/latex] and [latex]T_1 = 306 \text{K}[/latex], so that
[latex]\begin{array}{rl}\frac{Q}{t} &=\sigma eA(T_{2}^{4}-T_{1}^{4})\\ &=(5.67\times10^{-8}\text{J}/\text{s}\cdot \text{m}^2 \cdot \text{K}^4)(0.97)(1.50\text{m}^{2})[(295\text{K})^{4} - (306\text{K})^{4}]\\ &=-99\text{J}/\text{s} =-99\text{W}. \end{array}[/latex]
Significance
This value is a significant rate of heat transfer to the environment (note the minus sign), considering that a person at rest may produce energy at the rate of 125 W and that conduction and convection are also transferring energy to the environment. Indeed, we would probably expect this person to feel cold. Clothing significantly reduces heat transfer to the environment by all mechanisms, because clothing slows down both conduction and convection, and has a lower emissivity (especially if it is light-colored) than skin.
The average temperature of Earth is the subject of much current discussion. Earth is in radiative contact with both the Sun and dark space, so we cannot use the equation for an environment at a uniform temperature. Earth receives almost all its energy from radiation of the Sun and reflects some of it back into outer space. Conversely, dark space is very cold, about 3 K, so that Earth radiates energy into the dark sky. The rate of heat transfer from soil and grasses can be so rapid that frost may occur on clear summer evenings, even in warm latitudes.
The average temperature of Earth is determined by its energy balance. To a first approximation, it is the temperature at which Earth radiates heat to space as fast as it receives energy from the Sun.
An important parameter in calculating the temperature of Earth is its emissivity (e). On average, it is about 0.65, but calculation of this value is complicated by the great day-to-day variation in the highly reflective cloud coverage. Because clouds have lower emissivity than either oceans or land masses, they reflect some of the radiation back to the surface, greatly reducing heat transfer into dark space, just as they greatly reduce heat transfer into the atmosphere during the day. There is negative feedback (in which a change produces an effect that opposes that change) between clouds and heat transfer; higher temperatures evaporate more water to form more clouds, which reflect more radiation back into space, reducing the temperature.
The often-mentioned greenhouse effect is directly related to the variation of Earth’s emissivity with wavelength (Figure 1.33). The greenhouse effect is a natural phenomenon responsible for providing temperatures suitable for life on Earth and for making Venus unsuitable for human life. Most of the infrared radiation emitted from Earth is absorbed by carbon dioxide (CO2) and water (H2O) in the atmosphere and then re-radiated into outer space or back to Earth. Re-radiation back to Earth maintains its surface temperature about [latex]40^\circ \text{C}[/latex] higher than it would be if there were no atmosphere. (The glass walls and roof of a greenhouse increase the temperature inside by blocking convective heat losses, not radiative losses.)
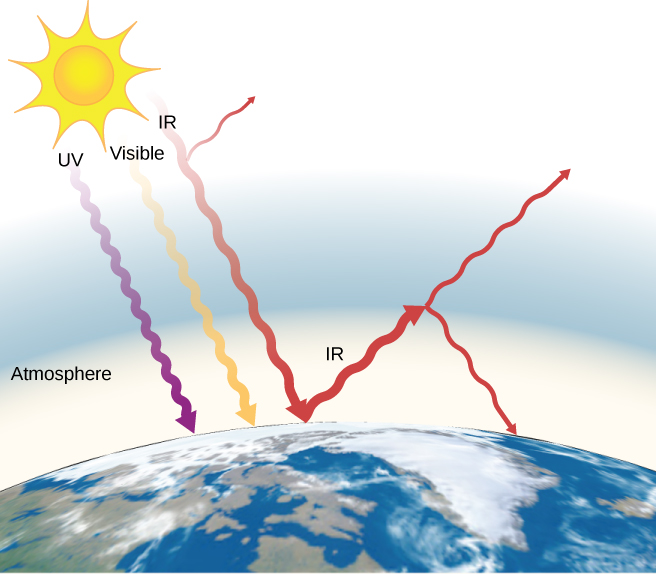
The greenhouse effect is central to the discussion of global warming due to emission of carbon dioxide and methane (and other greenhouse gases) into Earth’s atmosphere from industry, transportation, and farming. Changes in global climate could lead to more intense storms, precipitation changes (affecting agriculture), reduction in rain forest biodiversity, and rising sea levels.
Interactive
You can explore a simulation of the greenhouse effect that takes the point of view that the atmosphere scatters (redirects) infrared radiation rather than absorbing it and reradiating it. You may want to run the simulation first with no greenhouse gases in the atmosphere and then look at how adding greenhouse gases affects the infrared radiation from the Earth and the Earth’s temperature.
Problem-Solving Strategy: Effect of Heat Transfer
- Examine the situation to determine what type of heat transfer is involved.
- Identify the type(s) of heat transfer—conduction, convection, or radiation.
- Identify exactly what needs to be determined in the problem (identify the unknowns). A written list is useful.
- Make a list of what is given or what can be inferred from the problem as stated (identify the knowns).
- Solve the appropriate equation for the quantity to be determined (the unknown).
- For conduction, use the equation [latex]P = \frac{kA \Delta T}{d}[/latex]. Table 1.5 lists thermal conductivities. For convection, determine the amount of matter moved and the equation [latex]Q = mc \Delta T[/latex], along with [latex]Q = mL_\text{f}[/latex] or [latex]Q = mL_\text{V}[/latex] if a substance changes phase. For radiation, the equation [latex]P_\text{net} = \sigma eA(T_{2}^{4} - T_{1}^{4})[/latex] gives the net heat transfer rate.
- Substitute the knowns along with their units into the appropriate equation and obtain numerical solutions complete with units.
- Check the answer to see if it is reasonable. Does it make sense?
Check Your Understanding 1.9
How much greater is the rate of heat radiation when a body is at the temperature [latex]40^\circ \text{C}[/latex] than when it is at the temperature [latex]20^\circ \text{C}[/latex]?
Media Attributions
- Figure 1.19
- Figure 1.20
- Figure 1.21
- Figure 1.22
- Figure 1.23
- Figure 1.24
- Figure 1.25
- Figure 1.26
- Figure 1.27
- Figure 1.28
- Figure 1.29
- Figure 1.30
- Figure 1.31
- Figure 1.32
- Figure 1.33

